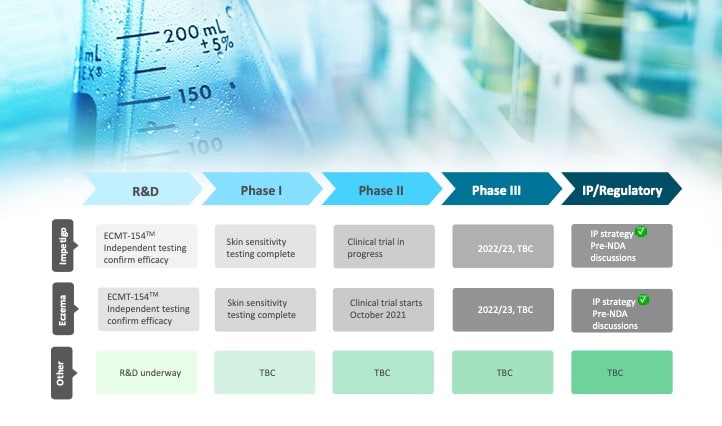
Clinical Trials
As an evidence-focused company, Manuka Bioscience is running clinical trials to demonstrate the efficacy of its mānuka oil based therapeutic products.
Eczema (Atopic Dermatitis) Clinical Trial
Eczema, also referred to as atopic dermatitis or atopic eczema, is a common chronic inflammatory skin disease characterized by recurrent flares of dry red and itchy skin lesions and subsequent remissions. Pathogenic manifestations of the condition include a defective skin barrier, recurrent infections, and local and systemic type 2 immune responses.
The condition has a considerable impact on quality of life because of skin discomfort and sleep disturbances. Eczema affects up to 30% of children and 10% of adults globally. There is a high prevalence of eczema in developed countries, and an increasing prevalence in developing countries, especially in urbanized areas.
Approximately 230 million people around the world have eczema and the condition does not yet have a permanent cure. Treatment options are limited and are accompanied by unpleasant, sometimes serious side effects.
During the Covid-19 pandemic, recommended strategies to minimise eczema flares included: reducing the usage of immunosuppressants such as steroids and maximising the use of moisturisers and topical anti-inflammatories and antimicrobial agents.
For these reasons, there is a is a significant opportunity to develop new, broad spectrum eczema treatments based on botanical ingredients that combine antimicrobial and anti inflammatory activity such as ECMT-154™.
In December 2021, Manuka Bioscience in partnership with Medical Research Institute of New Zealand will begin a stage II randomised, single blind, two arm eczema clinical trial with 118 patients aged 18-65 years.
This 12-month trial will compare the efficacy of the topical use of mānuka oil-based ECMT-154 TM cream with moisturiser-based vehicle control in treating moderate to severe eczema.
If you would like to participate in this trial, please contact gabby.shortt@mrinz.ac.nz
Clinical Trial Pipeline